Hydrobromic Acid
Hydrobromic Acid Specification
- Place of Origin
- India
- Moisture (%)
- Not specified (Aqueous solution)
- Molecular Formula
- HBr
- EINECS No
- 233-113-0
- Viscosity
- Approx. 2 mPas at 25C
- Residue on Ignition
- <0.01%
- Storage
- Store in tightly closed container, in a cool, dry, well-ventilated area away from incompatible substances
- Molecular Weight
- 80.91 g/mol
- Ph Level
- <1 (Very acidic)
- Assay
- Min 48% w/w
- Heavy Metal (%)
- <0.001%
- HS Code
- 28111940
- Boiling point
- 122C (252F)
- Particle Size
- Not applicable (liquid)
- Melting Point
- -11C (12.2F)
- Other Names
- Hydrogen bromide solution
- CAS No
- 10035-10-6
- Type
- Inorganic Acid
- Grade
- Reagent Grade, Technical Grade
- Usage
- Industrial, Laboratory, Pharmaceuticals, Organic synthesis, Catalysis
- Purity
- 48%
- Appearance
- Colorless to pale yellow liquid
- Application
- Pharmaceutical API intermediate, Bromination agent, Laboratory reagent, Chemical synthesis
- Raw Material
- Bromine, Water
- Smell
- Pungent, irritating odor
- Color
- Colorless or light yellow
- Form
- Liquid
- Corrosivity
- Corrosive to metals and tissue
- Flash Point
- Not flammable
- Packing Group
- II
- Density
- 1.49 g/cm at 20C
- Solubility
- Miscible with water, ethanol, ether
- Stability
- Stable under recommended storage conditions
- UN Number
- 1788
- Refractive Index (n20/D)
- 1.427
- Shelf Life
- 24 months in sealed container
- Transport Hazard Class
- 8 (Corrosive)
- Reactivity
- Reacts violently with bases and oxidizing agents
- Odor Threshold
- 0.2-1.0 ppm (as HBr vapor)
Hydrobromic Acid Trade Information
- Minimum Order Quantity
- 1 ,
- Payment Terms
- Letter of Credit (L/C), Paypal, Western Union, Cash in Advance (CID), Cash Advance (CA)
- Supply Ability
- 20 Per Week
- Delivery Time
- 7 Days
- Sample Available
- Yes
- Sample Policy
- Sample costs shipping and taxes has to be paid by the buyer
- Main Export Market(s)
- Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa, Asia
- Main Domestic Market
- All India
About Hydrobromic Acid
Hydrobromic Acid
Hydrobromic acid is a strong acid formed by dissolving the diatomic molecule hydrogen bromide (HBr) in water. "Constant boiling" hydrobromic acid is an aqueous solution that distills at 124.3 C and contains 47.6% HBr by weight, which is 8.89 mol/L. Hydrobromic acid has apKa of 9, making it a stronger acid than hydrochloric acid, but not as strong as hydroiodic acid. Hydrobromic acid is one of the strongest mineral acids known
Hydrobromic acid is mainly used for the production of inorganic bromides, especially the bromides of zinc, calcium, and sodium. It is a useful reagent for generatingorganobromine compounds. Certain ethers are cleaved with HBr. It also catalyzes alkylation reactions and the extraction of certain ores. Industrially significant organic compounds prepared from hydrobromic acid include allyl bromide, tetrabromobis(phenol), and bromoacetic acid.
Specification
| Packaging Type | HDPE Drum |
| Usage | Industrial |
| Packaging Size | 500-1000 kg |
| Physical State | Liquid |
Superior Reagent for Synthesis & Catalysis
Hydrobromic Acid plays a critical role in organic synthesis, bromination reactions, and as a catalyst in pharmaceutical and chemical manufacturing. Renowned for its high purity and reliable reactivity, this acid is frequently employed as a laboratory reagent and API intermediate, making it indispensable in advanced chemical processes.
Assured Quality and Safe Handling
Produced under stringent quality control, this product maintains a minimum 48% assay and complies with international standards for heavy metal and residue content. With a shelf life of 24 months when properly stored in tightly sealed containers, its stability and robust packaging ensure both safety and longevity in industrial settings.
Versatility Across Industries
With its high solubility and non-flammable nature, Hydrobromic Acid serves as a key raw material in multiple applications, from pharmaceutical synthesis to catalysis. Its adaptability and consistent performance make it suitable for laboratories, commercial manufacturing, and as an essential export product.
FAQs of Hydrobromic Acid:
Q: How should Hydrobromic Acid be safely stored and handled?
A: Hydrobromic Acid should be stored in tightly closed containers in a cool, dry, and well-ventilated area, away from incompatible substances such as bases and oxidizing agents. Use chemical-resistant gloves and eye protection when handling, as the acid is highly corrosive to metals and tissue.Q: What is the main industrial benefit of using Hydrobromic Acid (48%)?
A: Hydrobromic Acid is valued in industry for its ability to efficiently act as a bromination agent, facilitate organic synthesis, and serve as a catalyst in pharmaceutical and chemical production, thanks to its high purity and strong reactivity.Q: When is Hydrobromic Acid commonly utilized in the pharmaceutical sector?
A: Hydrobromic Acid is used as an API intermediate and in synthesis during the manufacture of various pharmaceuticals, especially when bromine incorporation or strong acid conditions are required in reactions.Q: Where is Hydrobromic Acid commonly supplied or exported from?
A: This Hydrobromic Acid is manufactured, exported, and supplied primarily from India, adhering to rigorous quality standards for global distribution.Q: What precautions should be taken during the transport of Hydrobromic Acid?
A: Hydrobromic Acid is classified as a Class 8 corrosive substance (UN 1788, Packing Group II). It must be transported in tightly sealed, compliant containers with clear hazard labeling by authorized carriers equipped for corrosive substances.Q: How can Hydrobromic Acid be used in laboratory or synthesis settings?
A: In laboratories, Hydrobromic Acid is used for bromination, as a catalyst, and for various chemical syntheses. Its strong reactivity under controlled conditions makes it effective for research and industrial-scale production.Q: What is the typical shelf life and purity of this Hydrobromic Acid?
A: The acid has a shelf life of 24 months when stored in a sealed container under proper conditions. Its purity is assured at a minimum of 48% w/w, with tight controls on residue and heavy metal content.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical Chemicals Category
Triethylene Triamine
Price 325 INR
Minimum Order Quantity : 25 Kilograms
Loss on Drying : 0.2%
Melting Point : 39C
Particle Size : Not applicable (liquid)
Smell : Other, Ammoniacal
Methane Sulfonyl Chloride
Price 43750 INR
Minimum Order Quantity : 1 Dram
Loss on Drying : 0.5%
Melting Point : 32 C
Particle Size : Not applicable (liquid)
Smell : Pungent
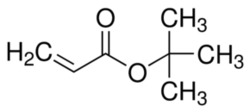
Butyl Acrylate
Price 500 INR
Minimum Order Quantity : 25
Loss on Drying : Not applicable (volatile liquid)
Melting Point : 64 C
Particle Size : Not applicable (liquid product)
Smell : Other, Fruity, pungent odor
Sodium Hydrosulphite
Price 10000 INR
Minimum Order Quantity : 10 Drams
Loss on Drying : 1.0% max
Melting Point : Decomposes below melting
Particle Size : Fine powder
Smell : Other, Slight sulfurous odor
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese Send Inquiry
Send Inquiry



 Send Inquiry
Send Inquiry Send SMS
Send SMS
