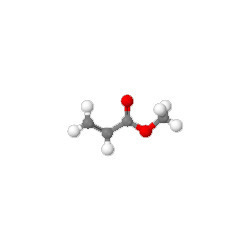
Dimethyl Carbonate
Dimethyl Carbonate Specification
- Assay
- 99.9%
- Loss on Drying
- 0.05%
- Residue on Ignition
- 0.001%
- Moisture (%)
- 0.01%
- Heavy Metal (%)
- 0.0001%
- Boiling point
- 90C
- Melting Point
- 2-4C
- Molecular Weight
- 90.08 g/mol
- Acid Value
- 0.01 mg KOH/g
- EINECS No
- 210-478-4
- Ph Level
- Neutral
- Particle Size
- Not Applicable (liquid)
- HS Code
- 29209020
- Storage
- Store in cool, dry place
- Place of Origin
- China
- Viscosity
- 0.585 mPas (at 25C)
- Molecular Formula
- C3H6O3
- Other Names
- DMC, Carbonic acid dimethyl ester
- CAS No
- 616-38-6
- Type
- Chemical Intermediate
- Grade
- Industrial Grade
- Usage
- Pharmaceuticals, Solvents, Polycarbonate Production
- Purity
- 99.9%
- Appearance
- Clear Liquid
- Application
- Electrolyte solvents, Methylating agent, Polycarbonate synthesis
- Raw Material
- Methanol, Carbon Monoxide
- Smell
- Faint, pleasant
- Color
- Colorless
- Form
- Liquid
- Solubility
- Miscible with organic solvents, slightly soluble in water
- Vapor Pressure
- 3.2 kPa (20°C)
- Auto Ignition Temperature
- 458°C
- Refractive Index
- 1.368 (20°C)
- Stability
- Stable under recommended storage conditions
- Flash Point
- 18°C (Closed cup)
- Density
- 1.069 g/cm³ (20°C)
Dimethyl Carbonate Trade Information
- Minimum Order Quantity
- 1 Dram
- Payment Terms
- Letter of Credit (L/C), Paypal, Western Union, Cash in Advance (CID), Cash Advance (CA)
- Supply Ability
- 10 Drams Per Week
- Delivery Time
- 7 Days
- Sample Available
- Yes
- Sample Policy
- Sample costs shipping and taxes has to be paid by the buyer
- Main Export Market(s)
- Eastern Europe, Central America, Middle East, South America, Western Europe, Australia, Asia, North America, Africa
- Main Domestic Market
- All India
About Dimethyl Carbonate
Dimethyl Carbonate
Dimethyl carbonate (DMC) is an organic compound with the formula OC(OCH3)2. It is a colourless, flammable liquid. It is classified as a carbonate ester. This compound has found use as a methylating agent and more recently as a solvent that is exempt from classification as a volatile organic compound (VOC) in the US. Dimethyl carbonate is often considered to be a green reagent.
Specification
| Density | 1.07 g/cm3 |
| Usage | Industrial |
| Physical State | Liquid |
| Grade Standard | Technical Grade |
High Purity and Versatility
Dimethyl Carbonate (DMC) offers a remarkable 99.9% purity, making it suitable for demanding industrial applications. Its versatility allows its use as a chemical intermediate for pharmaceuticals, as a solvent, and in polycarbonate synthesis. Its miscibility with organic solvents and neutral pH underscore its adaptability across several chemical processes.
Safe Handling and Storage Guidelines
DMC is a liquid with a low flash point, requiring cool, dry storage to ensure safety and stability. It remains stable under recommended conditions, with an auto-ignition temperature of 458C. Appropriate handling methods, such as closed containers and proper ventilation, help maintain its quality and minimize risks.
Environmental and Process Benefits
As a methylating agent and electrolyte solvent, DMC presents environmental advantages over traditional reagents due to its low toxicity and high efficiency. Its role in polycarbonate manufacturing also supports sustainable material development, delivering clean performance and minimal byproducts.
FAQs of Dimethyl Carbonate:
Q: How should Dimethyl Carbonate be stored to maintain its stability?
A: Dimethyl Carbonate should be stored in a cool, dry place away from ignition sources. Use tightly sealed containers to prevent moisture ingress, as the compound remains stable under recommended storage conditions.Q: What are the main industrial applications of Dimethyl Carbonate?
A: DMC is primarily used as a chemical intermediate in pharmaceuticals, a solvent, and in the synthesis of polycarbonates. Additional applications include use as a methylating agent and electrolyte solvent.Q: When is Dimethyl Carbonate most beneficial in chemical processes?
A: Dimethyl Carbonate is most beneficial when high purity and low toxicity are required, such as in pharmaceutical manufacturing, advanced materials synthesis, and eco-friendly solvent applications.Q: Where is Dimethyl Carbonate sourced and supplied from?
A: This chemical is commonly exported, imported, and supplied by manufacturers in China, with Indian companies acting as exporters, importers, and suppliers for industrial use.Q: What safety precautions should be taken during handling and use of Dimethyl Carbonate?
A: Due to its low flash point and vapor pressure, ensure the working area is well-ventilated, avoid heat sources, and wear suitable protective equipment such as gloves and goggles.Q: How does the use of Dimethyl Carbonate benefit polycarbonate production?
A: In polycarbonate synthesis, DMC enables efficient, high-yield production with fewer hazardous byproducts, contributing to more sustainable and safer manufacturing processes.Q: Can Dimethyl Carbonate be mixed with water and other solvents for formulation purposes?
A: DMC is miscible with most organic solvents but only slightly soluble in water. For optimal formulation, it is best combined with organic media rather than aqueous solutions.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharma Chemicals Category
Butylated Hydroxy Toluene
Price 300 INR
Minimum Order Quantity : 25 Kilograms
Purity : >99%
Storage : Other, Store in a cool, dry place, tightly closed container, protected from light
Form : Solid
Usage : Preservative, Antioxidant in food, cosmetics, pharmaceuticals, rubber, and petroleum products
Dichloromethane Chemical
Price 10340 INR
Minimum Order Quantity : 1 Dram
Purity : 99.9%
Storage : Other, Store in a cool, wellventilated place, tightly closed
Form : Liquid
Usage : Solvent, Paint remover, Metal cleaning, Pharmaceuticals
Liquid Methyl Acrylate
Price 120 INR
Minimum Order Quantity : 25 Kilograms
Purity : 99%
Storage : Room Temperature
Form : Other
Methane Sulphonyl Chloride
Price 175 INR
Minimum Order Quantity : 25 Kilograms
Purity : 99.5 %
Storage : Room Temperature
Form : Liquid
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese Send Inquiry
Send Inquiry


 Send Inquiry
Send Inquiry Send SMS
Send SMS
